There are a few updates that we want to share with you. First, Health Canada has just released an advisory regarding an EpiPen® manufacturing issue affecting a small quantity of devices.
Secondly, as some of you continue to have issues accessing your full prescription of epinephrine auto-injectors, we have provided recommendations on how you can obtain the devices. With a temporary second supplier in place (AUVI-Q®), everyone should be able to access auto-injectors. In fact, by having temporary supply, it’s our expectation that we get back to the recommended guidance of having two devices on hand. We’ll continue to advocate on this.
Health Canada public advisory notice
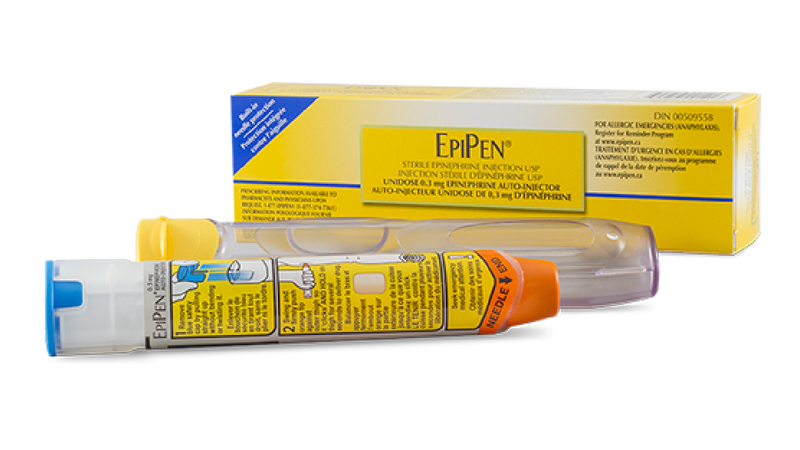
Health Canada has released a notice to advise that there is a manufacturing issue which affects a limited number of EpiPen and EpiPen Jr auto-injectors in Canada.
The issue with these devices is that they may not slide out of their carrier tube easily, which could delay or potentially prevent emergency treatment. The problem is with the device label, and not with the auto-injector itself. The devices impacted can be the EpiPen 0.3 mg or EpiPen Jr 0.15 mg doses with expiry dates ranging from April 2018 to October 2019.
Please read Pfizer Canada’s notice for further information. This is not a product recall.
How you can check to see if your device is affected
 Check the expiry dates of your EpiPens – impacted devices have an expiry date from April 2018 to October 2019.
Check the expiry dates of your EpiPens – impacted devices have an expiry date from April 2018 to October 2019.- Ensure your devices can be easily removed from the carrier tube.
 If you’re not sure if your devices have been impacted, ask your local pharmacist to examine them for you.
If you’re not sure if your devices have been impacted, ask your local pharmacist to examine them for you.- Request replacement devices if required.
Accessing epinephrine auto-injectors: follow these steps
As of September 7th, temporary AUVI-Q epinephrine auto-injectors were made available across pharmacies in Canada. To better understand your recent experiences in accessing devices since that time, we asked you to complete a quick survey. Thank you for providing your feedback!
 From the survey, you told us:
From the survey, you told us:
- You had challenges in filling your prescriptions for epinephrine auto-injectors
- Limits were placed on the number of devices you received, and some of you still weren’t able to access a device
- Pharmacists weren’t aware of AUVI-Q availability
- AUVI-Q coverage was limited by some private benefit plans (e.g. insurance companies, employers, etc.)
- Pharmacists asked you for a prescription for each device

Because of your feedback, we recommend the following steps to ensure you get access to epinephrine auto-injectors:
- Ask your pharmacist if epinephrine auto-injectors (e.g. EpiPen and AUVI-Q) are available in-store. If they aren’t available, provide them with the product identification numbers (e.g. DIN numbers) so the products can be ordered:
- EpiPen 0.3 mg: 00509558
- EpiPen Jr 0.15 mg: 00578657
- AUVI-Q 0.3 mg: 02480379
- AUVI-Q Jr 0.15 mg: 02480360
- If you have private insurance coverage, check with your insurance provider to see if all auto-injectors (e.g. EpiPen and AUVI-Q) are covered. If the devices are not covered, you can request coverage.
- If you have public/government coverage, most provinces are covering both EpiPen and AUVI-Q.
- You may be required to have a prescription for AUVI-Q. Ask your allergist or physician for a prescription that includes both EpiPen and AUVI-Q on the same prescription, so you don’t have to go back to your physician.
- Continue to follow our safety tips for managing food allergies and preventing reactions.
You can count on us to continue advocating for ongoing access to epinephrine auto-injectors on your behalf. With two suppliers currently in the market, everyone should be able to get devices. We will continue to work closely with Health Canada, alternate providers, and other key stakeholders on this issue.
For further information or questions regarding the public advisory, please contact Pfizer Canada directly at 1-877-EPIPEN1 (1-877-374-7361).
Tags: access to epinephrine, advisory, auvi-q, EpiPen